Your cart is empty
FREE expedited shipping on all orders over $50 at checkout.*
Start ShoppingBrowser not supported. Improve your experience by using Google Chrome, Mozilla Firefox, Apple Safari or Microsoft Edge.
FREE shipping at $35






This product is not eligible for purchase with a promo code.
Your health insurance company may be able to reimburse you for part of the cost of this test: learn more.
No returns accepted
You must meet requirements outlined by the CDC to take this test
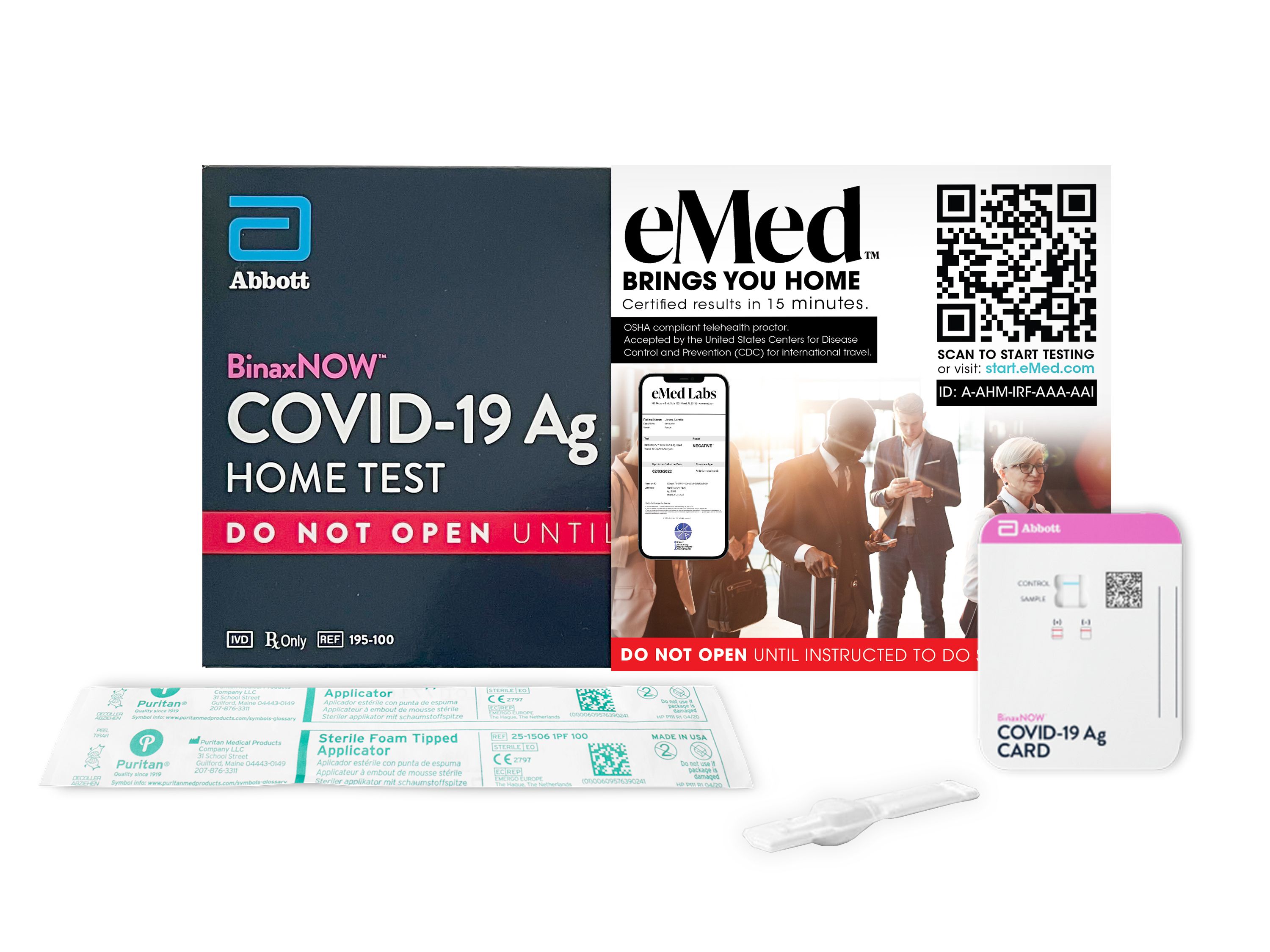
Abbott BinaxNOW™ COVID-19 Ag Card Home Test is FDA Emergency Use Authorized. This test combined with eMed telehealth services, and eMed lab's result, meets conditions of proof of negative COVID-19 test by the CDC. This is accepted by airlines for passengers traveling to countries that accept an antigen test for entry, and by cruise lines with ships originating from the US or Canada.
Abbott BinaxNOW™ COVID-19 Ag Card Home Test is FDA Emergency Use Authorized. This test combined with eMed telehealth services, and eMed lab's result, meets conditions of proof of negative COVID-19 test by the CDC. This is accepted by airlines for passengers traveling to countries that accept an antigen test for entry, and by cruise lines with ships originating from the US or Canada.
Your test kit includes a personal virtual visit with an eMed Certified Guide who will help you properly administer the test and third party verify the result. You must provide your official eMed Labs report in order to board your flight or cruise. The report is provided via email and can be printed or displayed on screen to provide proof.
**You must meet all requirements as outlined by the CDC to purchase this test. Click “Get Started” below to see if you qualify. Must be at least 18 years of age to order. Test authorized for use by persons age 2+. Test takers 14 or younger must be accompanied by an adult during specimen collection.
This item is HSA/FSA eligible.
ORDER YOUR KIT ONLINE Complete the required COVID-19 Testing Questionnaire by clicking “Get Started” to obtain a prescription to order tests.
TEST FROM HOME AT EMED.COM After you receive your test, visit eMed.com/app/start-testing to begin your virtual visit. An eMed Certified Guide will join you in a video visit to guide you through the testing process. Collecting a sample is an easy and gentle process, no different than using a cotton swab to clean the inside of your nose. See technical requirements for more details.
GETS RESULTS IN 15 MINUTES Test results will be made available to you right away from within the NAVICA app. When you receive your result, you will also receive a secure digital certificate in the NAVICA app. This certificate allows you to use your smartphone to store, display, and share your test results where applicable. It's important to remember the NAVICA digital certificate should not replace existing measures such as mask-wearing, social distancing, and frequent hand-washing.
WHAT HAPPENS IF I RECEIVE A POSITIVE RESULT? If you receive a positive result, you should remain in self-isolation, and inform your healthcare provider of your result. Always follow the guidance provided by the Centers for Disease Control and Prevention (CDC).
Warnings
The test does not replace the advice of a healthcare provider. You should seek immediate medical attention if you or your family members demonstrate emergency warning signs of COVID-19 (trouble breathing, bluish lips or face, or severe and constant pain or pressure in the chest). This list of symptoms is not all inclusive. Please consult your medical provider for any other symptoms that are severe or concerning.
The manufacturer of the BinaxNOW COVID-19 Ag Card Home Test Kit requires test kits be stored at temperatures between 2 and 30°C (35.6 - 86°F). Any extended exposure of the test kit to temperatures beyond this range may affect quality of test results. If you have ordered home delivery of the test kit and expect temperatures in your area to be outside of this range at time of delivery, it is recommended that you arrange for someone to be home at or near time of delivery to receive the test kit.
The BinaxNOW™ COVID-19 Ag Card Home Test has not been FDA cleared or approved. It has been authorized by the FDA under an emergency use authorization. The test has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens, and is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostic tests for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
SKU811877011330
Return PolicyThis item cannot be accepted
Product Weight2.50 OZ
Dimensions
Product Height5.00 in
Width8.25 in
Product Depth0.75 in