Your cart is empty
FREE expedited shipping on all orders over $50 at checkout.*
Start ShoppingBrowser not supported. Improve your experience by using Google Chrome, Mozilla Firefox, Apple Safari or Microsoft Edge.
FREE shipping at $35



Your health insurance company may be able to reimburse you for part of the cost of this test: learn more.
No returns accepted
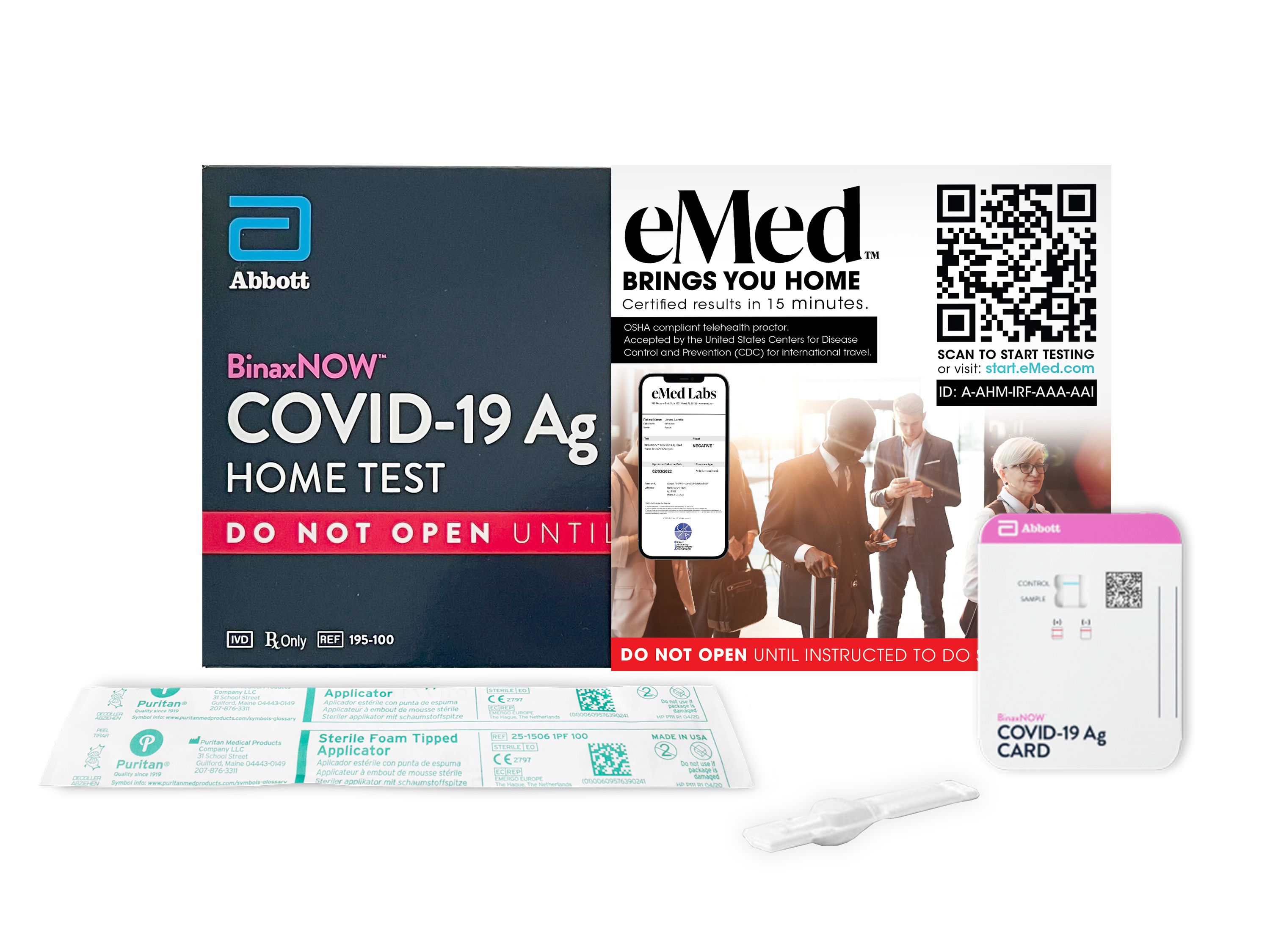
Abbott BinaxNOW™ COVID-19 Ag Card Home Test is FDA Emergency Use Authorized. This test combined with eMed telehealth services, and eMed lab's result, meets conditions of proof of negative COVID-19 test by the CDC. This is accepted by airlines for passengers traveling to countries that accept an antigen test for entry, and by cruise lines with ships originating from the US or Canada.
Abbott BinaxNOW™ COVID-19 Ag Card Home Test is FDA Emergency Use Authorized. This test combined with eMed telehealth services, and eMed lab's result, meets conditions of proof of negative COVID-19 test by the CDC. This is accepted by airlines for passengers traveling to countries that accept an antigen test for entry, and by cruise lines with ships originating from the US or Canada.
6-Pack includes six individually wrapped test, each test includes a personal virtual visit with an eMed Certified Guide who will help you properly administer the test and verify the result in just 15 minutes. Your eMed Labs result report is provided via email, as well as within the NAVICA app, and can be printed or displayed on your mobile device.
This item is HSA/FSA eligible.
When it's time to take your test, scan the QR code on the front of the box or visit eMed.com/app/start-testing to get started with your eMed virtual visit.
An eMed Certified Guide will take you through the test process, and you will have results in 15 minutes.
An eMed Lab Report with your test result will be delivered to your inbox.
If you receive a positive result, you should remain in self-isolation, and inform your healthcare provider of your result. Always follow the guidance provided by the Centers for Disease Control and Prevention (CDC).
Warnings
The manufacturer of the BinaxNOW COVID-19 Ag Card Home Test Kit requires test kits be stored at temperatures between 2 and 30°C (35.6 - 86°F). Any extended exposure of the test kit to temperatures beyond this range may affect quality of test results. If you have ordered home delivery of the test kit and expect temperatures in your area to be outside of this range at time of delivery, it is recommended that you arrange for someone to be home at or near time of delivery to receive the test kit.
The test does not replace the advice of a healthcare provider. You should seek immediate medical attention if you or your family members demonstrate emergency warning signs of COVID-19 (trouble breathing, bluish lips or face, or severe and constant pain or pressure in the chest). This list of symptoms is not all inclusive. Please consult your medical provider for any other symptoms that are severe or concerning.
The BinaxNOW™ COVID-19 Ag Card Home Test has not been FDA cleared or approved. It has been authorized by the FDA under an emergency use authorization. The test has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens, and is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostic tests for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
SKUUHGABEMED006
Return PolicyThis item cannot be accepted
Product Weight2.50 OZ