Your cart is empty
FREE expedited shipping on all orders over $50 at checkout.*
Start ShoppingBrowser not supported. Improve your experience by using Google Chrome, Mozilla Firefox, Apple Safari or Microsoft Edge.
FREE shipping at $35


The FDA has granted twenty-four (24) months of shelf-life at 2–30°C for On/Go™ One COVID-19 Antigen Home Test as of March 19, 2023. This new shelf-life extension represents an additional 3 months of shelf life, for a total twenty-four (24) months of total shelf life. Click here for more information.
No returns accepted
On/Go is the FDA-authorized, fast and easy-to-use COVID-19 self-test and companion app that guides you step-by-step through the testing process. In only 10 minutes, you will have the results you need to confidently return to work, school, sports and all the things you love to do, with the ability to store and share your family's and your test results from the intuitive On/Go app. Contains 1 test.
On/Go One COVID-19 Rapid Antigen Home Test. Authorized by FDA under an Emergency Use Authorization (EUA). Please download the free, optional companion mobile app for the best experience, available on both Apple and Android smartphones. On/Go One is your all in one test-to-treat COVID-19 solution.
Your health insurance company may be able to reimburse you for part of the cost of this test, learn more.
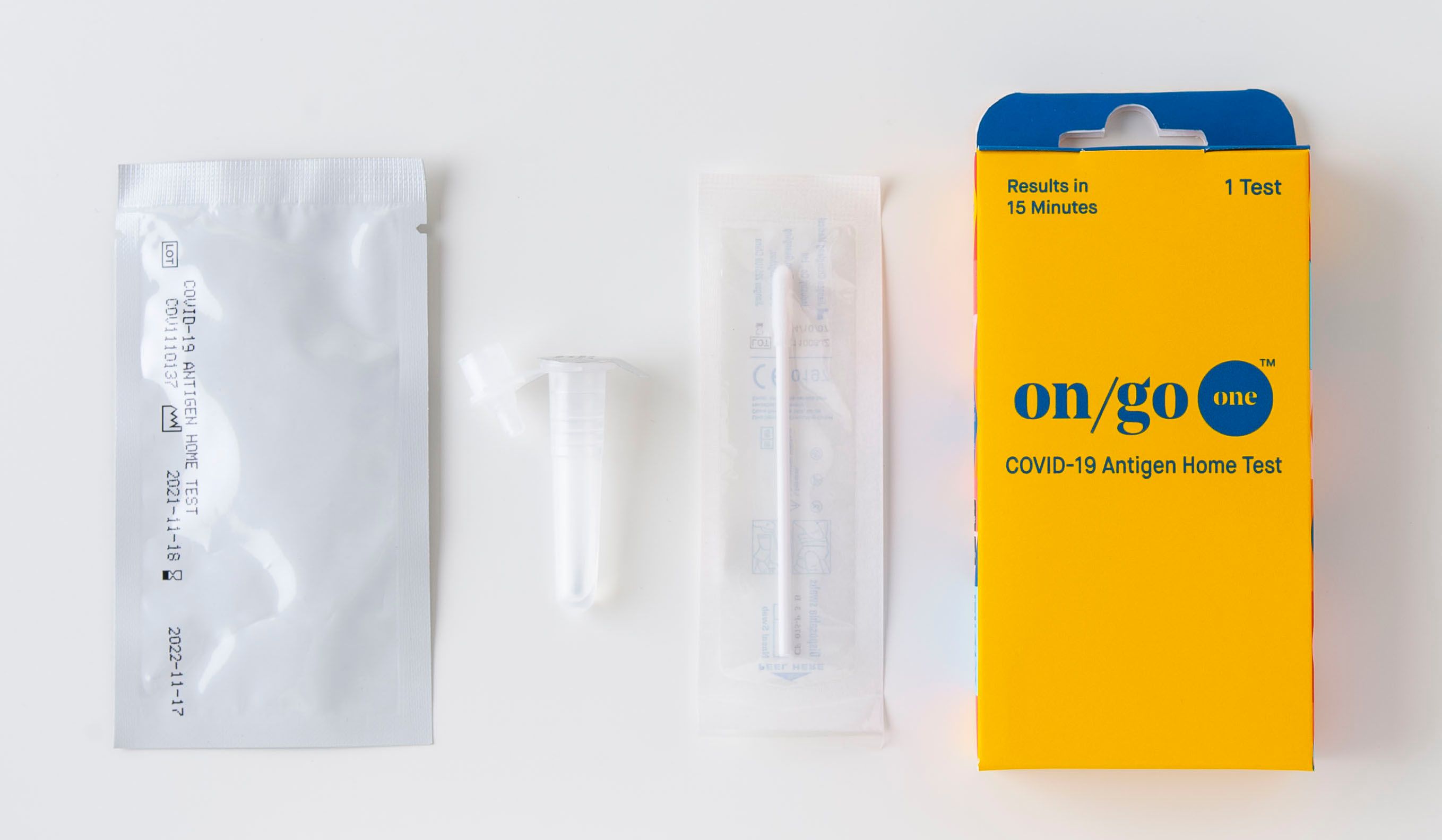
What's included
Contains: 1 plastic cassette, 1 swab, 1 buffer vial with cap, 1 set of instructions in English and Spanish
This item is HSA/FSA eligible.
This test is packaged with 1 test per box.
Contains: 1 plastic cassette, 1 swab, 1 buffer vial with cap, 1 set of instructions in English and Spanish
*PPA 93%/ NPA 100% see IFU for more details
Step-by-Step Instructions: The On/Go companion app, downloadable by scanning the QR code on box or available in the Apple App Store and Google Play, guides you seamlessly through each test step, from specimen collection to results interpretation.
Warnings
This product has not been FDA cleared or approved but has been authorized by FDA under an Emergency Use Authorization (EUA). This product has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens, and is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
SKU860007930409
Model NumberL031-118B3W
Return PolicyThis item cannot be accepted
Product Size1.00 CT
Product Weight1.00 OZ
Dimensions
Product Height5.30 in
Width2.50 in
Product Depth0.80 in